Need a Reliable Vancomycin Hydrochloride IV Infusion Manufacturer in India?

Meropenem Injection Manufacturing: The Key to Pharma Business Success
May 12, 2026In today’s healthcare industry, reliability is everything, especially when it comes to critical care injectable medicines. Hospitals, pharmaceutical distributors, healthcare providers, and procurement companies all depend on consistent access to safe and effective antibiotic infusions.
Among these life-saving medicines, Vancomycin Hydrochloride IV Infusion plays a vital role in treating severe bacterial infections and supporting patient care in emergency and hospital settings.
However, finding a dependable Vancomycin Hydrochloride IV Infusion manufacturer is not always simple. Many buyers face challenges such as inconsistent supply chains, poor-quality injectable products, delayed deliveries, and manufacturers who fail to meet international pharmaceutical standards.
That’s why choosing a trusted pharmaceutical manufacturing partner matters more than ever.
A reliable injectable medicine manufacturer not only ensures product quality but also provides advanced sterile manufacturing, regulatory compliance, timely delivery, and long-term business support.
Companies like Monark Criticare, Monark Biocare, and Fedley Healthcare are helping healthcare providers and pharmaceutical businesses access dependable Injectable Medicines & Critical Care solutions with high manufacturing standards and professional support.
Table of Contents
ToggleWhy Choosing the Right Vancomycin IV Infusion Manufacturer Matters
Choosing the right Vancomycin IV infusion manufacturer ensures sterile production, accurate dosing, and consistent quality, which are critical for patient safety and effective treatment of serious bacterial infections.
Reliable manufacturers also follow strict regulatory standards, maintain batch consistency, and ensure uninterrupted supply to hospitals, supporting healthcare providers in delivering safe and timely life-saving treatment; it’s about ensuring:
- Consistent product quality
- Safe sterile manufacturing
- Reliable pharmaceutical supply
- WHO-GMP compliance
- Proper batch testing and validation
- Long-term pharmaceutical partnership
A trusted manufacturer helps healthcare providers avoid supply interruptions while maintaining confidence in patient treatment outcomes.
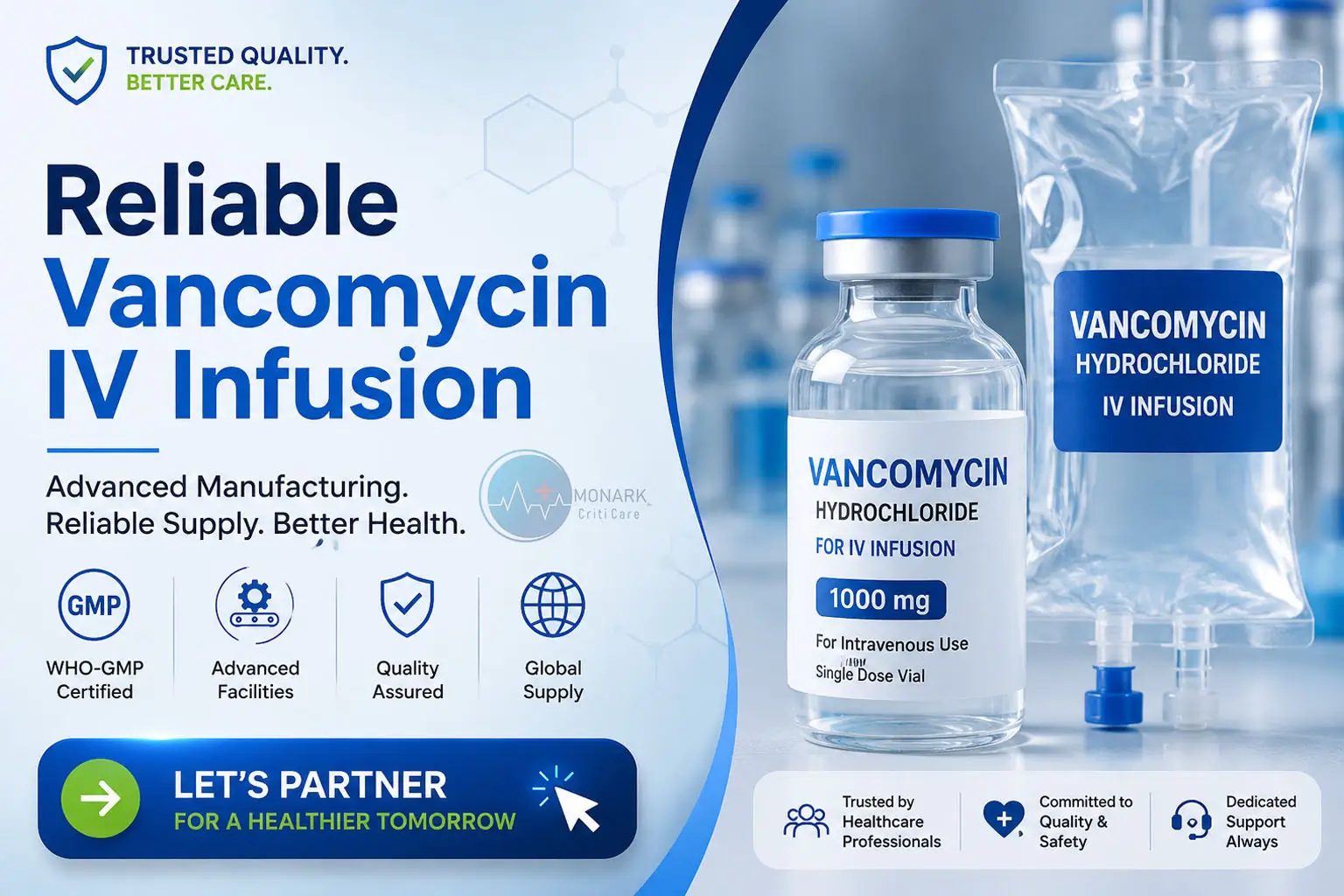
Partner with a trusted pharmaceutical manufacturer for high-quality Vancomycin Hydrochloride IV Infusion solutions.
Growing Demand for Injectable Medicines & Critical Care Products
The demand for Injectable Medicines & Critical Care products has increased significantly due to expanding healthcare infrastructure, rising hospital admissions, and growing global pharmaceutical demand.
Healthcare organizations now prefer manufacturers that offer:
- Advanced sterile injectable production
- Regulatory-compliant formulations
- Large-scale manufacturing capacity
- Efficient pharmaceutical logistics
- High-quality antibiotic infusions
This is where experienced pharmaceutical companies stand out in the market.
Businesses searching for dependable pharmaceutical manufacturing solutions often look for companies specializing in third-party manufacturing and critical care injectable production.
What Makes a Reliable Vancomycin Hydrochloride IV Infusion Manufacturer?
Not every pharmaceutical company follows the same production and quality standards. A reliable Vancomycin Hydrochloride IV Infusion manufacturer focuses on quality, consistency, compliance, and patient safety throughout the production process.
Here are some important qualities to consider:
1. WHO-GMP Certified Manufacturing Facilities
A WHO-GMP-certified pharmaceutical facility demonstrates that the company follows internationally recognized manufacturing standards.
This includes:
- Sterile production environments
- Controlled manufacturing systems
- Proper documentation practices
- Batch traceability
- Strict quality assurance protocols
When selecting a pharmaceutical partner, regulatory compliance should always be a top priority.
2. Advanced Sterile Injectable Infrastructure
Modern IV infusion manufacturing facilities use advanced cleanroom systems and automated equipment to ensure contamination-free production.
High-quality manufacturers invest in:
- Sterile filling lines
- Modern packaging systems
- Automated production technologies
- Environmental monitoring systems
- Product stability testing
This helps maintain consistent pharmaceutical quality across every batch.
3. Strong Pharmaceutical Quality Assurance
Reliable manufacturers conduct rigorous quality testing before releasing products into the market.
Essential quality measures include:
- Sterility testing
- Endotoxin analysis
- Stability testing
- Batch validation
- Product quality monitoring
Manufacturers with strong quality assurance systems build greater trust among hospitals, distributors, and healthcare organizations.
Benefits of Partnering with an Indian Pharmaceutical Manufacturer
India has become one of the world’s leading pharmaceutical manufacturing hubs. Global healthcare companies often rely on Indian manufacturers for cost-effective and high-quality injectable medicines.
Here’s why:
| Advantage | Benefit |
| Advanced Manufacturing | High-quality sterile production |
| Cost Efficiency | Competitive pricing |
| Skilled Workforce | Experienced pharma professionals |
| Export Capabilities | Reliable international supply |
| Regulatory Standards | WHO-GMP-compliant production |
Indian pharmaceutical manufacturers are widely recognized for delivering reliable healthcare solutions while maintaining international standards.
Reliable Third-Party Manufacturing Solutions
Many pharmaceutical companies and distributors prefer third-party manufacturing services to expand their product portfolios without building their own production facilities.
Benefits include:
- Reduced operational costs
- Faster product launch
- Scalable production capacity
- Professional manufacturing support
- Regulatory compliance assistance
This business model allows companies to focus on branding, marketing, and distribution while experienced manufacturers handle production.
Manufacturers offering third-party manufacturing often provide specialized support for critical care injections and antibiotic infusions.
Business Growth Through PCD Franchise Services
Business Growth Through PCD Franchise Services is rapidly rising in the pharmaceutical industry, offering strong opportunities for distributors and entrepreneurs to build scalable ventures with low investment and trusted product portfolios from established healthcare companies.
Through PCD Franchise Services, partners gain monopoly rights, marketing support, and consistent supply, enabling faster business expansion, higher profitability, and long-term growth in the competitive pharmaceutical market.
Benefits include:
- Monopoly rights opportunities
- Marketing and promotional support
- Access to quality pharmaceutical products
- Brand development assistance
- Long-term business growth potential
Many businesses prefer partnerships with established pharmaceutical manufacturers because of their strong market reputation and dependable supply systems.
Understanding the ANCOLASE Product Range
Understanding the ANCOLASE product range helps healthcare businesses access trusted antibiotic injectable solutions designed for hospital-grade treatment, ensuring quality, safety, and therapeutic effectiveness for critical care applications and patient management.
Explore Monark Criticare’s Third Party Manufacturing and PCD Franchise services to get reliable pharmaceutical support, consistent production, and a wide ANCOLASE range tailored for distributors, hospitals, and healthcare providers seeking dependable supply solutions.
Products such as:
reflect a manufacturer’s ability to support critical care treatment protocols with quality-focused pharmaceutical solutions.
A broad product portfolio also demonstrates strong manufacturing capability and healthcare industry expertise.
Why Hospitals Prefer Trusted Injectable Manufacturers
Hospitals prefer trusted injectable manufacturers because they ensure consistent product quality, strict regulatory compliance, and reliable supply chain continuity. These manufacturers reduce treatment risks, support emergency care needs, and help healthcare institutions maintain safe, effective, and uninterrupted patient care for critical conditions.
Here are some key reasons:
- Consistent Product Availability: Hospitals require an uninterrupted medicine supply to avoid treatment delays and maintain smooth patient care operations.
- International Pharmaceutical Standards: Manufacturers following international compliance standards help healthcare providers maintain confidence in patient safety and treatment quality.
- Efficient Distribution Networks: Reliable pharmaceutical logistics systems ensure timely delivery to hospitals, distributors, and healthcare organizations.
- Professional Technical Support: Trusted manufacturers provide regulatory guidance, documentation support, and product-related assistance for business partners.
Choosing a trusted injectable manufacturer ultimately helps healthcare providers maintain operational efficiency, strengthen patient care standards, and ensure long-term pharmaceutical supply reliability.
How to Evaluate a Vancomycin Hydrochloride IV Infusion Supplier
Before selecting a pharmaceutical manufacturing partner, consider the following factors:
| Evaluation Factor | What to Check |
| Certifications | WHO-GMP & ISO compliance |
| Manufacturing Facility | Sterile injectable infrastructure |
| Product Portfolio | Critical care and antibiotic products |
| Quality Control | Batch testing systems |
| Production Capacity | Bulk order capability |
| Export Support | Global distribution network |
Careful evaluation helps buyers identify manufacturers capable of supporting long-term healthcare and business goals.
Importance of Vancomycin Hydrochloride 1000 MG in Critical Care
Vancomycin Hydrochloride 1000 MG is essential in critical care units for treating life-threatening bacterial infections. It delivers strong antibacterial action, helping clinicians control resistant pathogens quickly and effectively, supporting improved patient outcomes in urgent hospital treatment situations.
Since this medicine is commonly used in critical care settings, manufacturers must maintain:
- High sterility standards
- Accurate dosage formulation
- Secure pharmaceutical packaging
- Reliable product quality validation
- Consistent manufacturing processes
This highlights the importance of partnering with a dependable pharmaceutical manufacturer.
Why Monark Criticare Stands Out
Monark Criticare has built a strong reputation in Injectable Medicines & Critical Care manufacturing by focusing on product quality, advanced infrastructure, and customer-focused pharmaceutical solutions.
The company supports healthcare providers and pharmaceutical businesses with:
- Advanced injectable manufacturing
- Critical care product expertise
- Quality-driven production systems
- Reliable Third-Party Manufacturing support
- Professional pharmaceutical services
Its focus on quality, compliance, and consistency makes it a preferred pharmaceutical partner.
Conclusion
Finding a reliable Vancomycin Hydrochloride IV Infusion manufacturer is essential for healthcare providers, pharmaceutical distributors, and hospitals seeking quality, consistency, and long-term reliability.
From advanced sterile production and WHO-GMP-certified facilities to scalable manufacturing and global distribution support, trusted pharmaceutical companies play a crucial role in modern healthcare systems.
If you are looking for a trusted partner for Vancomycin Hydrochloride 1000 MG, Third Party Manufacturing, or PCD Franchise Services, partnering with an experienced pharmaceutical company can help ensure long-term business success and reliable healthcare delivery.
Frequently Asked Questions
1. What is Vancomycin Hydrochloride IV Infusion used for?
It is commonly used in hospitals and critical care settings to treat severe bacterial infections requiring intravenous antibiotic therapy.
2. Why is WHO-GMP certification important?
WHO-GMP certification ensures that pharmaceutical products are manufactured under strict safety and quality standards.
3. What should I look for in a reliable IV infusion manufacturer?
You should evaluate:
- Regulatory certifications
- Sterile manufacturing capabilities
- Quality assurance systems
- Supply reliability
- Pharmaceutical industry experience
4. Why are Indian pharmaceutical manufacturers globally trusted?
Indian pharmaceutical companies are recognized for advanced manufacturing infrastructure, competitive pricing, skilled professionals, and strong export capabilities.